Gene drives: Current assessments not tailored
Submitted by huib_lis on
The National Instute for Public Health and the Environment (RIVM) of the Netherlands, resorting under the Ministry of Health, Welfare and Sport, recently published a Policy Report on gene Drives.
An analysis by RIVM indicates that current methods for assessing the risks for human health and the environment are less suitable for the effects of gene drives. RIVM recommends that for applications in laboratories of organisms with a gene drive, an authorization should be obligatory. A notification is insufficient.
Legally, an organism with a gene drive is a genetically modified organism (GMO), for which an authorisation or notification is mandatory in the Netherlands. Working with GMOs is only allowed if risk assessment shows that the risks for human health and the environment are negligible.
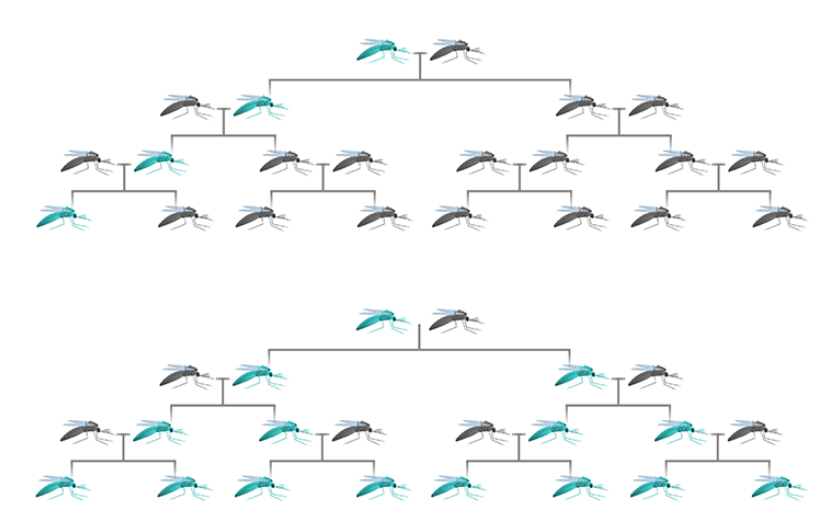
The current assessment method is not or partially tailored to GMOs with a gene drive, because the rapid and permanent alteration of the entire population is insufficiently taken into account. Sporadically, a gene drive may occur by accident when researchers apply genetic components in such a way that they inadvertently form a gene drive.
RIVM further recommends adapting current legislation in such a way that it is no longer possible to inadvertently create a gene drive. In addition, authorisation may only be granted if the required information is available to answer all questions in the risk assessment. This secures a safe application of organisms with a gene drive and provides opportunity to gain knowledge about a gene drives functioning and its effects. Finally, an international approach is desirable, because it may involve organisms and possible effects on human health and the environment, which could spread across national borders.
The entire report (in English) can be dowloaded here
Image derived from Harvard University – Gene Drives FAQ
